The Complete Code of Federal Regulations, Title 21, Food And Drugs, FDA Regulations, 2016 - Kindle edition by United States Government. Professional & Technical Kindle eBooks @ Amazon.com.
.jpg)
FDA CFR Title 21 Food and Drugs Regulations - TELUGU GMP - Provides GMP Pharmaceutical Guidelines in Telugu.

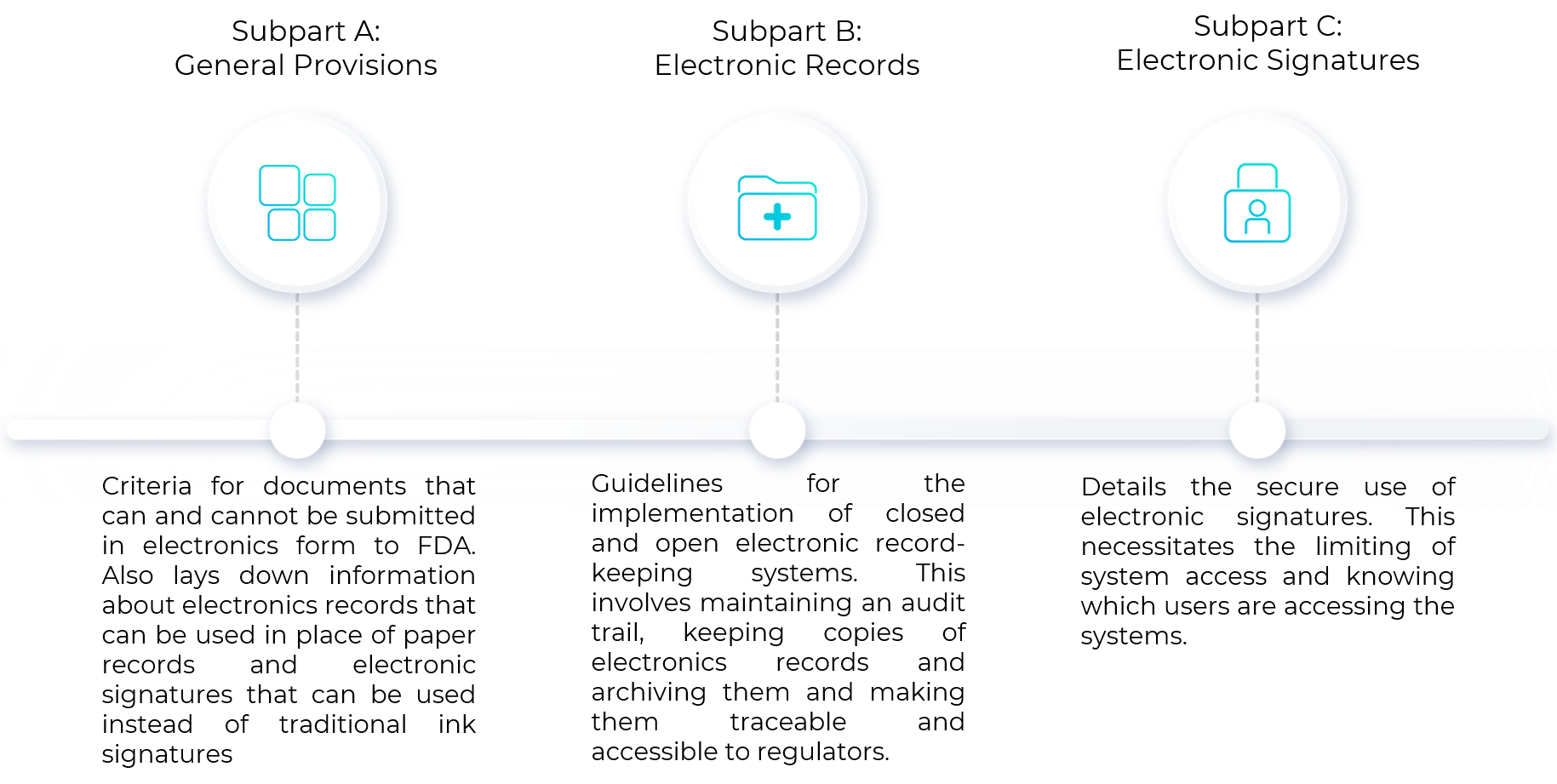
Book 23: 2022 Part 11 & Drug Development: Regulation, Preamble & FDA G – Clinical Research Resources, LLC
.jpg)
How many titles are there in the FDA CFR (Code of Federal Regulations)? - TELUGU GMP - Provides GMP Pharmaceutical Guidelines in Telugu.